Researchers at the University of Tsukuba in Japan have developed an all solid state magnesium air rechargeable battery that outperforms systems using platinum based cathodes. The design relies on a nitrogen doped nanoporous graphene structure that addresses a long standing engineering challenge which has limited the real world use of magnesium air batteries. The findings were published in the Advanced Functional Materials.
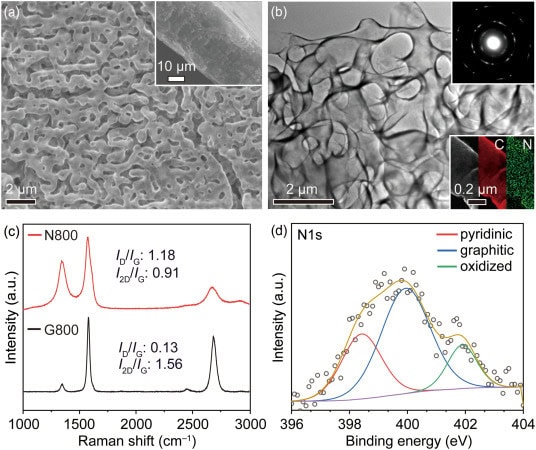
The battery uses commercially available magnesium metal as the anode and a polymer gel containing magnesium chloride as the solid electrolyte. The key innovation lies in the cathode. Researchers created a free standing three dimensional nanoporous graphene structure doped with nitrogen atoms. This design gives the cathode the catalytic properties and structural stability needed to function under the harsh electrochemical conditions inside a magnesium air battery.
Magnesium air rechargeable batteries use a carbon based cathode, a magnesium metal anode, and an electrolyte containing magnesium chloride. The system draws oxygen from the air as an active material at the cathode. This allows the construction of high capacity batteries using relatively inexpensive materials. Theoretical performance of magnesium air batteries approaches that of lithium air batteries. Magnesium also remains more abundant and cheaper than lithium and does not form dangerous dendrites during charging.
Durability has long limited the technology. Chloride ions inside the battery often trigger internal chlorination, which damages the cathode and shortens battery lifespan. Earlier solutions relied on platinum based cathodes. Those designs proved costly and impractical for large scale manufacturing.
The nitrogen doped graphene cathode changes this dynamic. By embedding nitrogen atoms inside a three dimensional nanoporous graphene framework, researchers produced a cathode with strong catalytic activity and improved structural resilience. The porous structure supports oxygen reduction reactions while allowing discharge products to move through the material more easily. This helps maintain stable electrochemical performance across repeated charging cycles.
The result is a cathode that resists chloride damage without relying on precious metals. This directly addresses cost and supply chain concerns associated with lithium based battery technologies.
The solid state design also improves safety. Using a solid electrolyte eliminates the leakage risks associated with liquid electrolyte batteries and improves mechanical flexibility. Tests showed the battery maintained its performance even when bent to an angle of 120 degrees without any electrolyte leakage.
The combination of flexibility and safety opens potential applications beyond electric vehicles. Possible uses include wearable electronics, flexible devices, and other technologies where rigid battery designs limit product development.
Magnesium air batteries have a typical energy density of about 6.8 kilowatt hours per kilogram and a theoretical operating voltage of 3.1 volts. Magnesium’s natural resistance to dendrite formation reduces safety risks often associated with lithium based batteries. Researchers view this chemistry as a promising long term alternative in advanced energy storage systems.
Global demand for solid state battery technology continues to grow as electrification expands across transportation, electronics, and grid energy storage. Materials that remain abundant and cost efficient will play a central role in future battery supply chains.
The next step for the research team involves demonstrating that the nanoporous graphene cathode can be manufactured at industrial scale. That milestone will determine whether the laboratory breakthrough moves toward commercial battery production.
You can read the research paper here.